Particulate contamination in drug formulations continues to be a cause for concern in the manufacture of injectable drugs. Despite extensive control and inspection during manufacturing, foreign particulate is a leading cause of injectable drug recalls.1-4 During the period 2008–2012, 22% of recalls for sterile injectable drugs were due to the presence of visible particles.2 An industry wide survey from 2014 indicates that glass particles are commonly found during visual inspection.2,3 These recalls represent a significant cost and liability to drug manufacturers and any emerging technologies that can help to mitigate the root cause of particulate generation can provide significant value, as well as a positive impact on patient safety.1,3-10
Glass particles are generated from a number of different mechanisms during the drug filling process. Vial breakage on filling lines is a generally well understood mechanism of particle generation and contamination. A less understood mechanism is the particle generation that results from glass-to-glass contact between vials during the typical bulk filling operation. Recent research published in the PDA Journal has identified and studied two new mechanisms of particulate generation. In the article entitled “Particulate Generation Mechanisms during Bulk Filling and Mitigation via New Glass Vial”, Corning scientists determined significant particulate generation was the result of frictive sliding contact and impact events between vials on filling lines. Through a robust understanding of the root cause, it is possible to explore new materials and methods that substantially reduce this mechanism of glass particle contamination, thereby mitigating a risk for drug products.
Frictive sliding
During drug filling operations, glass vials are in constant sliding contact with one another (Fig 1). The friction between the vials can generate significant scratches on vial surfaces, which in turn ejects glass particles. This process is known as frictive sliding contact.
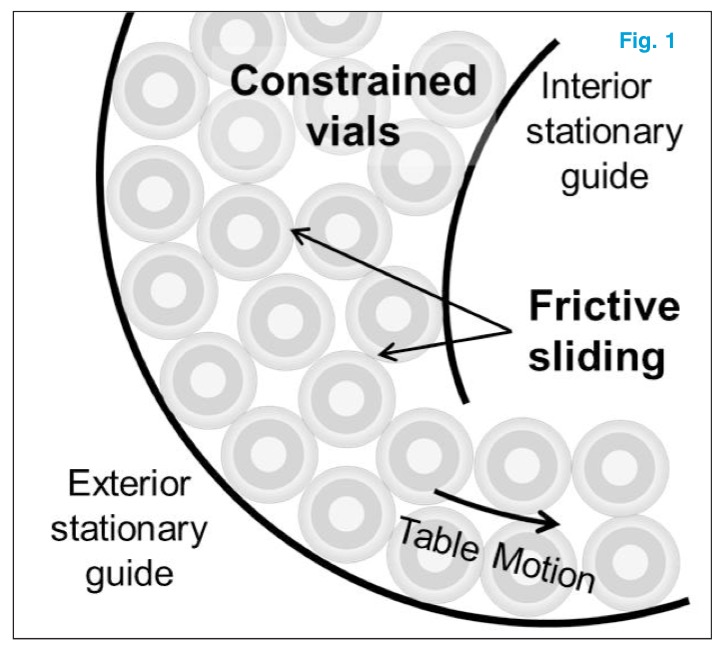
Vials in motion slide past vials constrained by the inner guide on a rotary accumulator table.
To better understand its impact, a laboratory method was developed to simulate glass vial frictive sliding contact. In this method, two glass vials are oriented orthogonally in a fixture with contact between the vial side walls. A mechanical tester applies a controlled, constant load and translates one of the vials linearly. During the scratch generation, glass particles are ejected to a nearby particle debris field. The debris field is microscopically inspected and the glass particles automatically counted using vision software (Fig. 2).

Exemplary debris field generated during frictive sliding simulation at 10 N. The area outlined in white is analysed by particle counting software
Conventional 3 ml borosilicate glass vials were tested with an applied load ranging from 1 to 30 N over a 1 mm scratch length. As would be expected, as the load is increased, the quantity of particles increased but the distribution was unaffected. Loads as low as 1 N generated a substantial quantity of particles (see Fig. 3). This lab test illustrates how frictive sliding contact, which is ubiquitous on traditional bulk sterile filling lines, is a significant source of glass particle generation.
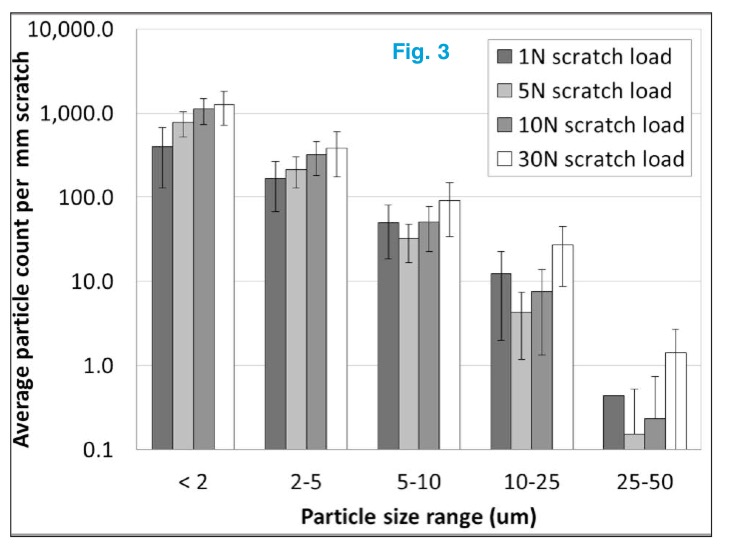
Quantification of particle size as result of different test loads
Impact events
In addition to frictive sliding contact, impacts between vials in manufacturing lines can also be a significant source of particulate generation.
Impact events are especially frequent when moving vials are accelerated towards stationary vials, as is the case when a screw feeder pushes vials into one another on a manufacturing line (Fig. 4).
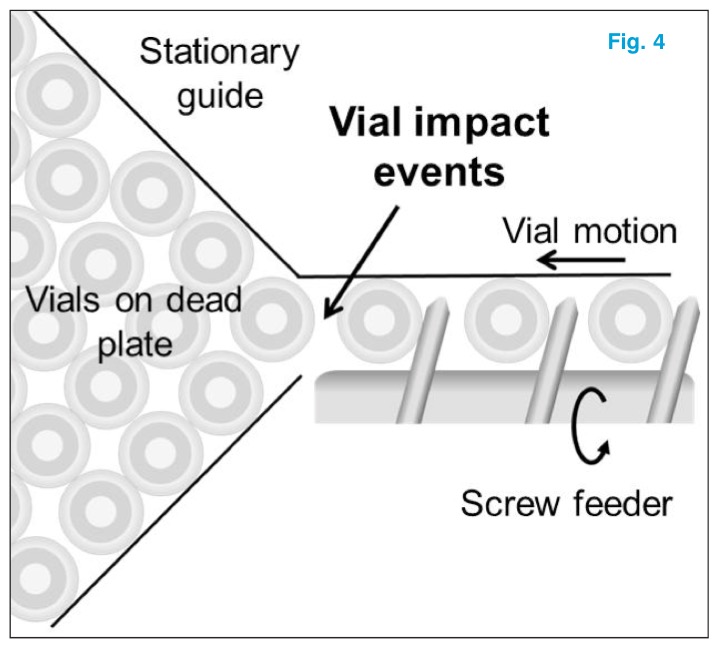
A screw feeder accelerates causing impact of vials into stationary vials on a dead plate
To study particle generation from impact events, a dynamic test system was developed that enables a single vial to be accelerated towards a constrained stationary vial at a controlled velocity. Impact velocities ranged from 400–1000 mm/sec representing an upper range of instantaneous speeds observed on the filling line.
As anticipated, increasing the surface stress and energy of the impact increases the severity of damage and frequency of checks. In general, impact of undamaged surfaces resulted in particles <100 µm (sub-visible classification); however, when damaged surfaces were impacted, particles >100 µm (visible classification) were observed.
In some instances (Fig. 5), a visible particle was generated and ejected from the interior surface of the vial upon impact. This particular type of impact event could result in contamination of the product and could occur after inspection.

Although the frequency of visible glass particle generation depends on many factors, including the line design, setup and operation, glass damage and impact events are unavoidable in current bulk filling operations, posing a risk of particulate generation by this mechanism.
New glass lowers peak particle counts
With a deep understanding of the mechanisms that lead to particulate matter in glass vials, a new vial was designed to substantially reduce glass particulate generation on filling lines. This vial leverages existing manufacturing and engineering platforms used to produce well-known glass technologies such as optical fibres, mobile phone glass, etc. The new vial is made from an aluminosilicate glass composition and strengthened, which provides superior strength, reducing the propensity for breakage and chipping. Additionally, the vial includes a low coefficient-of-friction (COF) exterior surface to reduce damage from frictive sliding contact.
Initial testing of the new vial was performed with above mentioned laboratory tests to evaluate performance during frictive sliding and impact interactions, compared with conventional borosilicate vials. Evaluation in both showed no observable glass damage or particle generation (Fig. 6).

Following laboratory evaluation of the new vial, a filling line trial was undertaken to quantify the particle reduction benefit in the filling line environment. This trial comprised of processing around one million 3 ml vials on a high-speed restricted access barrier style filling line with an experimental protocol specifically assessing airborne particulate matter response. The line was operated using typical production protocols and setpoints with an operating speed range of 500–550 vials/min. The results were compared with a conventional glass vial, run immediately before the new vial, using the same protocol and a similar quantity of vials. Table I shows a comparison of particulate performance metrics for the trial.
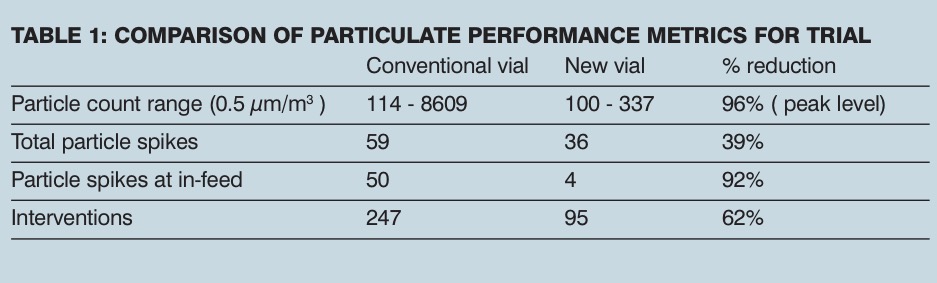
Particulate matter monitoring showed a 96% reduction in peak particle level (>0.5 µm particles) above the post-depyrogenation accumulator table, where vials are open and at risk to foreign matter contamination. This result demonstrated that by mitigating the generation mechanism, airborne particle levels can be substantially reduced during the filling process.
Additionally, while processing conventional glass vials on the filling line, frequent operator interventions were required to alleviate jammed, tipped or broken vials or to clean specific contain guide surfaces. When running the new glass vials with the low-COF surface, the machinability improved significantly, as evidenced by a 61% reduction in interventions and a 36% increase in effective line throughput.
Glass for pharma manufacturing
Glass-to-glass contact of conventional vials on filling lines is a substantial source of particulate matter. This contact significantly increases the likelihood of generating glass particles and is a cause of large quantities of subvisible glass particles. Drug manufacturers have an obligation to identify, understand and control critical process factors11,12 and to implement equipment, controls, protocols and inspections to reduce the risk of foreign particulate matter in conventional vials. However, these steps do not fully address the root cause of the problem.
A low COF external surface presents a solution to inherent manufacturing challenges. The new vial is designed to address a primary cause of both visible and sub-visible glass particulate matter generation during the filling process. Line trials have demonstrated a substantial reduction in both airborne particulate generations and operator interventions, presenting significant benefit to pharmaceutical manufacturers. They have also confirmed compatibility with existing manufacturing equipment and processes.
This study has demonstrated that, by understanding a cause of a major foreign particulate matter source, a low-COF external surface can have a significant, positive impact on product quality and safety.
References
- Langille, S. Visual Inspection of Injectables, PDA Visual Inspection Forum. Oct. 2015
- Tawde, S. (2014) J. Pharmacovigil. 3 (1)
- Langille, S. (2013) PDA J. Pharma. Sci. Tech. 67 186-200
- Bukofzer, S. et al. (2015) PDA J. Pharm. Sci. Technol. 69 123-129
- Gould, C. Lessons learned from glass recalls. PDA / FDA Glass Quality Conference. June 2012
- Jack, T. et al. (2010) Intensive Care Med. 36 707-711
- Walpot, H. et al.(1989) Anesthetist 38 544-548
- Zabir A. et al. (2008) Southern African Journal of Anesthesia and Analgesia 14 17-19
- Lehr, H. et al. (2002) Am. J. Respir. Crit. Care Med. 165 514-520
- Schroeder, H. et al. (1976) Am. J. Hosp. Pharm. 33 543-546
- Guidance for Industry: Sterile Drug Products by Aseptic Processing – Current Good Manufacturing Practice, USDHHS, FDA, CDER, CBER, ORA, Sep. 2004
- Guidance for Industry: Q9 Quality Risk Management, USDHHA, FDA, CDER, CBER, Sep. 2006
This article appeared in the February issue (2018) of Cleanroom Technology and is based on that written by Timmons, C. L., Chi, Y. L. and Merkle, S. (2017) first published in PDA J. Pharm. Sci. Technol. Vol. 71 5 pp379-392
