In 2015, CROs, CMOs, and increasingly CDMOs have all been playing larger roles in the development and manufacturing of pharmaceutical and biopharmaceutical products as drug manufacturers face rising cost and regulatory pressures. Healthy growth rates of approximately 9% and 6% CAGR for the contract research and manufacturing markets respectively reflect this trend.
Even so, competition has been stiffer for service providers as sponsor firms continue to pare down their vendor numbers and establish strategic partnerships with fewer, integrated suppliers. In addition, access to advanced capabilities and differentiating technologies and methodologies has become a fundamental driver for outsourcing and a major factor in the selection process. All of these trends are expected to continue to influence the contract services market into 2016.
Increased outsourcing activity
The strong growth rates for the pharmaceutical contract services market reflect increased spending on the part of both pharmaceutical and biopharmaceutical companies, as revealed by Nice Insight’s 2015 Pharmaceutical and Biotechnology Outsourcing Survey. While the percentage of survey participants whose companies spend more than US$50m on outsourcing has remained fairly stable over the last three years at 23–24%, the proportion of respondents whose companies spend $10–50m on outsourcing has increased dramatically from 38% to 62%, while the percentage of participants whose companies spend less than $10m has decreased by slightly more than half. Furthermore, regardless of the buyer group or budget size, the average number of services outsourced by survey participant companies increased from 2014 to 2015.
Changing dynamics in the industry are leading to this increased reliance on service providers. The drugs themselves – both small- and large-molecule – are more complex. Formulation to achieve high bioavailability, efficacy and safety has become more challenging. The shift to specialised medicines such as orphan drugs and therapies with breakthrough or fast track status has required the development and commercialisation of these complex products in half the normal time.
The growing complexity and cost of clinical trials have drawn the attention of the US Congress (21st Century Cures). Demonstration of real value to payers, physicians and patients, each of whom may have their own definitions for the term, has become imperative. Extension of product lifetimes through modification of delivery and/or packaging technologies has become crucial for achieving reasonable profitability levels given heightened generics competition. Outsourcing is an efficient, cost-effective way to meet many of these rapidly changing industry needs.
Growing importance of biopharma
Biopharmaceutical companies are increasingly turning to service providers for all aspects of drug development, partly to avoid the very high capital expenditure and long lead times needed to construct, equip and validate manufacturing facilities. In fact, the average number of services used by the survey respondents increased the most for those working in biotech (4.9% to 8.2%) and emerging biotech (5.4% to 9%) companies.
Large and emerging biotech companies are now outsourcing nearly as frequently as Big Pharma
In addition, large and emerging biotech companies are now outsourcing nearly as frequently as Big Pharma and generally more often than emerging pharma companies at the discovery, preclinical, and Phase I stages of drug development.
The percentage of survey participants whose companies are engaged in the development of biologics also increased from 65% in 2013 to 82% in 2015, while the percentage of outsourcing budgets spent on biologics vs small molecule therapeutics rose from 54% to 58% over the same period. These numbers reflect the growing importance of biopharmaceuticals to the overall drug industry.
More interest in emerging markets
The results of the Nice Insight survey also indicate that the growth in demand for contract services is a global trend, with many providers in emerging markets benefiting. Just over 81% of respondents indicated that they would consider outsourcing to CROs/CMOs in emerging markets, while only approximately 53% (52.73%) have previously worked with service providers in these regions.
There has been particularly strong growth in outsourcing by biopharmaceutical companies to CROs in emerging markets. In 2014, only 43% of survey participants reported outsourcing to these markets; that number climbed dramatically to 63% in the 2015 survey. CROs in emerging markets such as China, Eastern Europe, Turkey, Argentina, and Brazil offer biopharmaceutical manufacturers access to large, affordable talent pools with nearly comparable technical capabilities and skills combined with the potential to reduce R&D costs and development times.
There are concerns with outsourcing to emerging market CROs and/or CMOs. Of the small number (12%) of survey participants that aren’t interested in doing so, more than half (57%) cited uncertainty about quality levels and more than a third (36%) believe that the logistics are too complicated. Other concerns relate to regulatory compliance (29%), intellectual property issues (14%) and communications challenges (14%).
The impact of strategic partnerships
The growing prevalence of contract development and manufacturing organisations (CDMOs) is an important trend in 2015 that reflects the growing need of drug manufacturers for greater support throughout all phases of drug development and commercialisation. It also underscores the recent trend towards the formation of strategic partnerships with a few preferred suppliers that have the expertise to support fully integrated capabilities and culture, systems and processes necessary for the development of collaborative relationships. This approach allows drug manufacturers to meet aggressive development times while realising greater efficiencies in their own supply chains.
The growing prevalence of contract development and manufacturing organisations reflects the growing need of drug manufacturers for greater support through all phases of drug development and commercialisation
One consequence of this trend has been an increase in consolidation among CROs and CMOs as they attempt to become integrated CDMOs. For instance, both Patheon and Catalent announced IPOs in 2015 to raise capital for further expansions following significant acquisitions in 2014: Patheon bought DSM’s pharmaceutical products business and Gallus Biopharmaceuticals, and Catalent purchased Pharmapak Technologies, Redwood Bioscience and Micron Technologies. Other examples included the merger of Cambridge Major Laboratories with AAIPharma, the acquisition of Hameln Pharma by Siegfried Group, the purchase of Aesica Pharmaceuticals by Consort Medical, and very recently the acquisition of Coldstream Laboratories by Piramal Enterprises. LabCorp’s acquisition of Covance and Chiltern’s purchase of Theorem Clinical Research are examples of consolidation on the clinical side.
These activities demonstrate that the CDMO concept has become a reality. Of course, only time will tell if the companies that ultimately emerge will indeed be able to provide the fully integrated services and form the strategic partnerships demanded by their pharmaceutical and biopharmaceutical clients.
According to the Nice Insight survey results, sponsor companies look for strategic partners that have the capability and willingness to collaboratively develop operating procedures, use dedicated project managers, demonstrate a clear willingness to make long-term commitments and have the ability to customise protocols for different projects.
Before service providers can establish collaborative, long-term relationships with sponsor companies, they need to attract the attention of potential clients. The survey found that approximately two-thirds (67%) of companies in the pharmaceutical industry (big pharma, speciality/emerging pharma, biopharma, emerging biotech, medical/device and generics/biosimilars) use industry research to select service providers.
Matchmaking routes
Hiring consultants to find the most suitable match for their needs (59%) and referrals from colleagues (54%) ranked second and third, respectively. Attendance at industry trade shows and events (40%) and publications (36%) are also used, but are regarded as less important or less reliable tools in the selection process. Use of online directories (24%) and web searches (18%) were the least preferred screening methods.
CROs, CMOs and CDMOs that attract the attention of sponsor companies have a track record of success and financial stability and an industry reputation for doing quality work. They also have the necessary operational, methodological and therapeutic experience to meet a wide range of project needs and the ability to be adaptable and flexible.
Clearly demonstrated communication skills and a willingness to be transparent are also important to survey respondents, as is a deep understanding of customer needs. A reputation for being responsive, willing to go the extra mile to ensure success, implementing sponsor methodologies, and eagerness to foster good rapport among project team members will also draw the attention of drug companies looking for service providers.
Pressures from consumers, investors, insurance companies, regulators and governments to drive down costs and improve product performance (efficacy, ease of use, patient adherence, etc.) are driving the need to increase efficiency and productivity while reducing costs and development timelines. Not surprisingly, drug manufacturers are turning to new technologies that improve performance without affecting the quality, safety and efficacy of their final drugs products. Advanced technologies are helping to improve manufacturing processes, increase the sensitivity and accuracy of analytical systems and enhance productivity and efficiency across all activities from R&D to distribution.
Technology differentiates
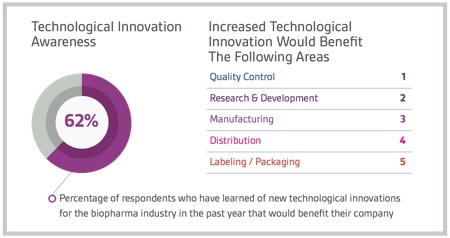
Introducing innovative new technologies to the lab, manufacturing plant and supply chains is in fact helping service providers attract projects from sponsors looking to be first to market with differentiated products. In fact, a high percentage of respondents (96%) indicated that they have at least some interest in forming outsourcing partnerships with service providers that adopt state-of-the-art technologies to increase efficiency, safety, quality and traceability.
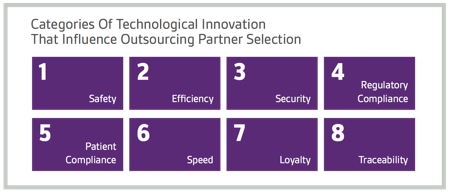
In addition, the level of technological innovation at a potential outsourcing partner influences the selection process for survey respondents. The use of advanced technologies to enhance safety was seen as most important, followed by improving efficiency, security, regulatory compliance, patient compliance, speed, loyalty and traceability.
The use of robotics labs to perform routine tests is an important mechanism for achieving cost and/or time savings when outsourcing to CROs
Survey respondents also ranked quality control, research and development, manufacturing, distribution, and labelling and packaging as having the greatest to least potential to benefit from technological innovation. These results suggest that the impact of technology on patient-centric traits is more important than its impact on business-related activities. They also indicate that CROs, CMOs and CDMOs all have the opportunity to capture new business and establish new partnerships by leveraging innovative technologies and methodologies.
With respect to particular technologies of interest to sponsor companies, half of the survey respondents indicated that the use of robotics labs to perform routine tests is an important mechanism for achieving cost and/or time savings when outsourcing to contract research organisations (CROs), second only to the use of cloud-based management services (62%).
A similar percentage of respondents from biotech and pharma companies indicated that cloud-based management services will be important for increasing cost and time efficiencies (63% and 61% respectively). That is not the case for the use of robotics; more survey participants from biotech companies (56%) than from traditional pharmaceutical companies (46%) believe that this technology will provide cost and time savings. This difference may reflect the fact that many small-molecule analyses have already been automated, or perhaps the greater complexity and longer timelines required for biologic samples.
Quality still matters
For the third year in a row, quality and reliability remain the top two priorities for sponsor companies when looking for outsourcing partners. Poor quality also remains the top source of dissatisfaction for survey respondents, followed by a lack of timeliness in resolving problems and unexpected charges.
Meanwhile, productivity continues to increase in importance for survey participants. It has steadily moved up in the rankings from fifth to third place from 2013 to 2015, probably reflecting the need for pharmaceutical and biotechnology companies to increase efficiencies and lower costs across all activities.
Looking forward
According to the Nice Insight survey, in the next year respondents are most likely to seek support for their clinical research, analytical, biomanufacturing, biostatistics, packaging and data management activities.
Increasing reliance on service providers does not guarantee success for all players in the market
Outsourcing is clearly growing in importance for the pharmaceutical industry, but this increasing reliance on service providers does not guarantee success for all players in the market. Service providers with a proven track record for quality and on-time delivery, access to advanced technologies, the ability to apply them to meet specific customer needs and a willingness to participate in long-term, collaborative relationships will have the greatest opportunity to benefit.
Not all CROs and CMOs, and even many CDMOs, will be able to meet the growing demands of pharmaceutical industry clients. It will be interesting to see how this dynamic situation plays out over the coming years.
Survey methodology
The Nice Insight Pharmaceutical and Biotechnology Survey is deployed to outsourcing-facing pharmaceutical and biotechnology executives on an annual basis. The 2014–2015 report includes responses from 2,303 participants. The survey is comprised of 240+ questions and randomly presents ~35 questions to each respondent in order to collect baseline information with respect to customer awareness and customer perceptions of the top ~125 CMOs and ~75 CROs servicing the drug development cycle.
Five levels of awareness, from ‘I’ve never heard of them’ to ‘I’ve worked with them’ factor into the overall customer awareness score. The customer perception score is based on six drivers in outsourcing: Quality, Innovation, Regulatory Track Record, Affordability, Productivity, and Reliability. In addition to measuring customer awareness and perception information on specific companies, the survey collects data on general outsourcing practices and preferences as well as barriers to strategic partnerships among buyers of outsourced services.
If you would like to learn more about Nice Insight, the report, or how to participate, please contact Guy Tiene by sending an email to guy@thatsnice.com.
