Despite the falling returns on investment in research and development experienced by most pharma companies in recent years, R&D spending is expected to grow by more than 8% in the next year, according to the latest CPhI Pharma Insights report analysing trends and technologies. Having moved away from single, large-scale R&D sites, the industry has diversified its interests geographically and technologically and increased collaboration and partnerships with smaller biotech companies and contract research organisations.
This approach is leading to increased open innovation, out-licensing and in-licensing of technology and outsourcing models that drive down development costs, such as partnering and profit-sharing arrangements, the report says.
Unsurprisingly, oncology continues to be the dominant focus, with almost a quarter of respondents to the survey putting anticancer treatments at the centre of their R&D efforts.
Unsurprisingly, oncology continues to be the dominant focus
The research divides into two clear areas, according to the report: inhibiting tumour progression and metastatis; and tumour shrinkage. 'Looking to the future, perhaps we will witness the two branches coming together – a sign of which is evident in a current clinical trial being carried out by a big-name pharma company,' the report suggests. 'This study of early stage anticancer treatment addresses the initial causes of the disease and is looking towards the development of novel therapeutics, such as kinase inhibitors, epigenome-modulating compounds and immunotherapies.'
There are currently an estimated 150 kinase-targeted drugs in clinical development out of approximately 1000 anticancer drugs being developed at present, it adds.
Joint-second most popular R&D areas are CNS, cardiovascular and antibiotics, targeted by 12%, 12% and 13% of respondents respectively. Given the predicted epidemic of diabetes, it is perhaps surprising that only 5% of respondents said their focus was in the area of R&D. 'Is this reflective of a lack of promising targets or the success of existing drugs in the market and therefore a reduced potential market share for newer, more expensive drugs?' the report asks. On the other hand, the number of companies working on new antibiotics is reassuring in view of the pressing need in the face of increasing bacterial resistance and despite their relatively low income potential.
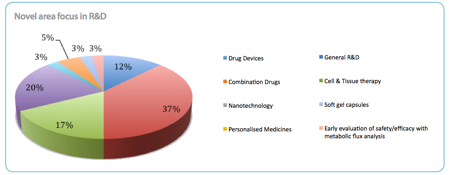
Combination drugs and personalised medicines are emerging as key novel areas for R&D, with 37% of respondents focusing on the former and 20% on the latter. 'Combination drugs and personalised medicines are complementary and it is likely that we will see the two go hand-in-hand more as industry looks to evolve within the oncology field,' the report predicts.
The oncology area is increasingly focused on cytotoxic and highly potent APIs, and antibody drug conjugates (ADCs) are increasingly coming to the fore in a trend that is likely to continue over the next few years. Manufacturing and R&D facilities will therefore need to have specialist skills to handle high potency compounds and controlled drugs.
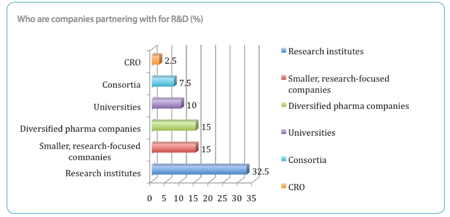
Decentralising away from Big Pharma and downsizing into a greater number of smaller and more nimble biotech, CRO and discovery and development companies has given the industry greater flexibility and the ability to switch between innovative targets, the report says. More than three-quarters of respondents are now partnering in their R&D programmes and nearly two-thirds are using a contract service providers such as a CRO or CDMO. The rise in partnerships is a reflection of the increasing need to reduce costs and ensures better manufacturing and development practices, the report points out.
The proportion of respondents partnering mainly with research institutes is 32.5%, with 15% working with diversified pharma companies and a further 15% with smaller, research-focused companies, suggesting that R&D activity has moved towards finding pre-existing targets with development potential rather than discovering their own NCEs.
Pharma companies and suppliers should be open about their respective skill sets to ensure that regulation and good manufacturing practice are not compromised
A key differentiator in the future of the R&D market, according to the report, will be access to the most innovative techniques and processes allowing previously unstable and/or difficult to formulate products with poor release profiles to come to market. Technologies that enable quicker development timelines, reduced costs and/or regulatory approvals are also keenly sought in a globally competitive marketplace.
'A progressive trend over the past 10 years has been the gradual shift of knowledge from Big Pharma to the CRO and outsourced market,' says the report. 'It is apparent that pharma companies and suppliers should be open about their respective skill sets to ensure that regulation and good manufacturing practice are not compromised.'
Selecting the right partners or outsourcing suppliers is more important than ever, and this concern is reflected in the fact that knowledge management and ensuring regulatory compliance were seen as major R&D challenges by 30% and 32.5% of respondents respectively. This has given rise to a new area of outsourced specialisms to ensure that projects are not held up by regulatory hurdles.
'What we are now seeing is the rise of the specialist within outsourced partners – a skill set to navigate the approvals process and QP release is vital in keeping R&D on time and on budget,' the report states.
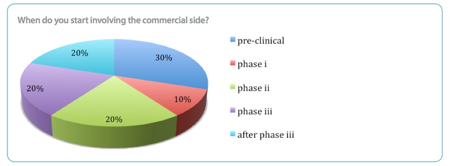
Improving efficiency was cited by almost 40% of respondents as a top challenge. Streamlining R&D and adopting processes and mechanisms to drive down costs and improve time to market are strategies being widely adopted, but these measures are not yet enough to overcome the challenge posed by competition from India and China where R&D costs are comparatively low. 'The quest to get drugs to market faster and reduce product failures and costly manufacturing corrections has understandably resulted in companies bringing in commercial side considerations at an earlier and earlier stage,' the report says.
Nearly a third of those surveyed were taking commercial considerations into account as early as the preclinical stage. In contrast, a disappointingly high proportion – a fifth – were not evaluating these until after Phase III.
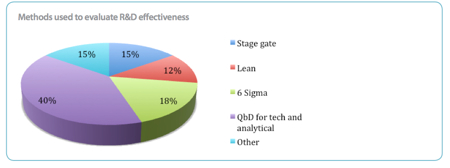
Another approach to improving R&D productivity is taking steps to quantify the value of the output. Tools like Process Analytical Technology (PAT) and Quality by Design (QbD) are being employed increasingly across the board, with 40% using QBD to measure both technical and analytical effectiveness. Six Sigma, Stage Gate and Lean are also used by 18%, 15% and 12% of respondents.
'Ensuring drug product quality at this earlier stage will be invaluable in meeting regulatory requirements during the approvals process, particularly for those in generics as a Questions-Based Review approach is now required by the FDA's Office of Generic Drugs,' the report points out.
It says that the Stage Gate approach has been particularly effective in analysing whether to continue financing a preclinical project into Phase I, but it is less clear whether it is an effective model for evaluating success at Phases II and III. Six Sigma and Lean are particularly effective at reducing waste when used in combination, but respondents expressed some concern that this approach may curtail true innovation.
The report concludes that there is a clear desire among all pharma companies to ensure that future R&D spending is effective and focused on bringing products to market. It predicts that batch processing will continue to give way to continuous processing at all phases of the development cycle and into full-scale manufacturing.
With the priority now given to cost-effective innovation models, it is no surprise that there is a more open innovation approach to technology licensing
With the priority now given to cost-effective innovation models, it is no surprise that there is a more open innovation approach to technology licensing, it says, with more than half of all respondents out-licensing their technologies and more than 60% using in-licensed technologies in their R&D efforts.
'This is an encouraging trend for the industry and one that, in the longer term, should provide increased cross-pollination of projects and enable even the smallest R&D companies access to technologies that could bring their products to market faster and, crucially, with a reduced R&D spend,' it concludes.
'With product development processes being increasingly managed through quality and control methods, the cost of and access to technology will be the essential access point on many projects ultimate viability.'
