This is most often oil particles dispersed in an aqueous phase, termed an oil-in-water emulsion, though water-in-oil emulsions are also common.
The majority of emulsions are thermodynamically unstable, and tend to break down over time via creaming, flocculation, Ostwald ripening, phase inversion and coalescence [Figure 1].
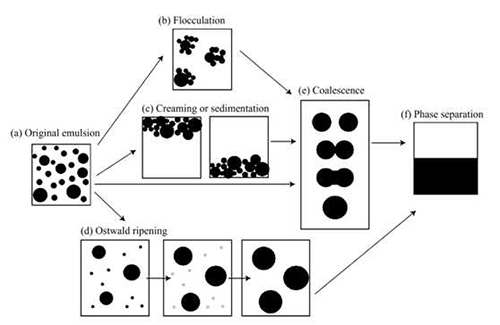
Figure 1: The different processes involved in the breakdown of an unstable emulsion
In cases of food or pharmaceutical products, it is important to minimise these processes, and maintain stability to facilitate the longest possible shelf life.
The droplet size in the dispersed phase can affect the stability of the emulsion.
Generally, the smaller the droplet size, the more stable the emulsion. More energy input is required to achieve smaller droplets and so high pressure homogenisation is often utilised to meet this, due to the high shear it can generate, to reduce droplet sizes to less than 1 µm.
Biopharma is known as a leading supplier of equipment to the pharmaceutical, biotech and process industries specialising in freeze drying, solvent removal/evaporation, high pressure homogenisation technologies and the related equipment from preparative chromatography systems to vial washers.
The Avestin range of high pressure homogenisers allows adjustable pressures of up to 30,000 psi to be induced upon a sample, as it is forced through a narrow nozzle orifice within the unique, dynamic homogenising valve.
As it exits the orifice, shear, cavitation and turbulence disrupts the particles, reducing their size.
It is important to note that higher energy input is not the only factor that determines final droplet sizes. Performing multiple passes through the homogenising valve can have benefits in not only reducing particle sizes further, but in narrowing the overall droplet size distribution.
Avestin systems are often utilised to create a loop, more so at the pilot and production scale, to allow for efficient multiple-pass processing. This is particularly advantageous in the systems which provide a constant flow-through capacity, independent of the induced homogenising pressure.
The GMP-suitable Avestin range, ranging from the benchtop (3 L/hr) scale to the production (1000 L/hr) scale, is widely implemented across numerous industries for emulsification, though other applications include cell disruption, particle size reduction (e.g API) and liposome sizing/encapsulation.
At the pilot and production scale, Avestin’s systems utilise a triplex pump design, whereby the pumps work intermittently to facilitate negligible pressure pulsation, and high throughput. This can aid in producing a more stable emulsion, with reduced wear on impact parts within the product pathway.
These large scale units are commonly implemented across the biotech and pharmaceutical industries, due to their robustness, scalability and GMP-suitability, making them an efficient choice at the manufacturing scale.

