The growth in biological and highly potent pharmaceutical formulations presents particular challenges for vial filling and handling.
SCHOTT Pharmaceutical Systems, a market leader in primary packaging made of glass and polymers, has developed a new generation of pharmaceutical vials; it’s designed to address the challenges of chemical interactions, glass particle contamination and the risk of breakage owing to the stresses of handling and transportation.
Created as a modular concept, the EVERIC family currently consists of three products that provide pharmaceutical companies with a unique combination of attributes.
Next-generation vials
More than two thirds of all drugs in the pipeline of pharmaceutical companies are biologics — and a large number will be highly potent products that need to be filled in volumes of less than 1 mL.
In such low-fill scenarios, the high ratio of wetted container surface to drug volume leads to a proportional increase in exposure to leachables.
Furthermore, the near-heel region of standard vials often acquires a non-homogenous chemical structure during the forming process and is prone to ion exchange, which might result in delamination.
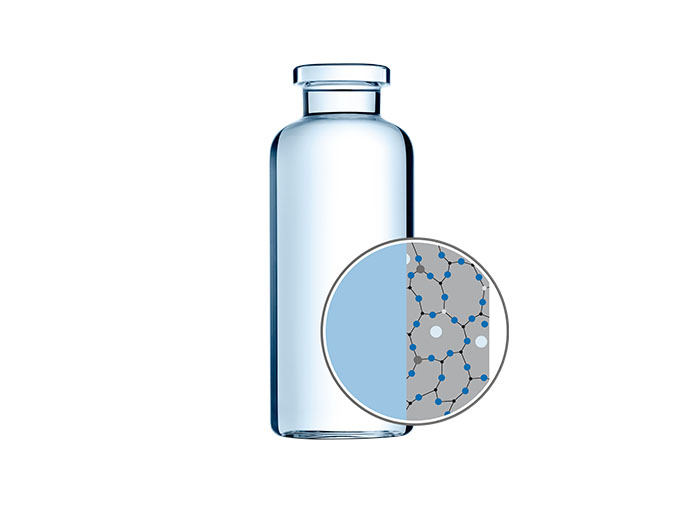
EVERIC pure
To counteract this, SCHOTT has developed EVERIC pure vials. Thanks to their improved chemical resistance, these vials show a very low concentration of leached “glass” elements even for low-fill applications.
The use of improved borosilicate glass tubing, FIOLAX CHR (controlled hydrolytic resistance), ensures higher chemical stability and leaves the original glass composition unchanged.
Manufactured on dedicated production lines in the US (Lebanon, Pennsylvania) and Germany (Müllheim), the vials are converted by SCHOTT’s approved delamination controlled (DC) forming process, which carries the chemical stability and homogenous surface of the glass tubing through the converting process.
Existing regulatory validation means that pharma companies can replace conventional tubular type-I glass vials with EVERIC pure without costly reregistration.
Strong and tolerant
A glass vial is only as strong as its weakest point. Defects such as scratches, cracks or flaking can occur during transport or on the filling line.
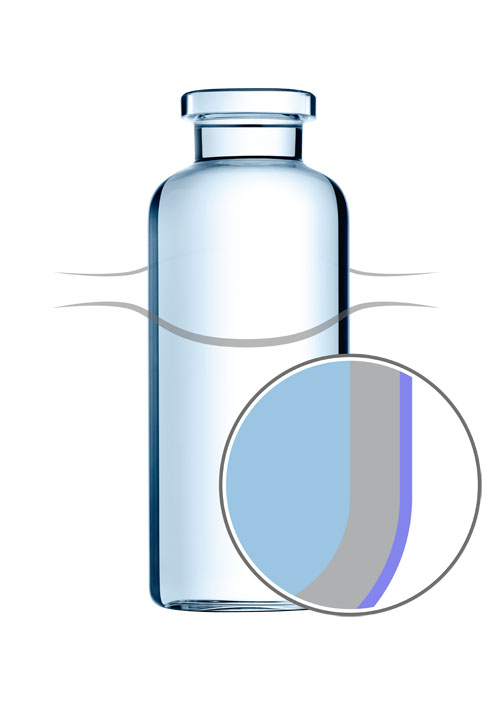
EVERIC smooth
With EVERIC strong and EVERIC smooth, SCHOTT offers a viable alternative to conventional glass vials and other vial types.. To create EVERIC strong, SCHOTT used computer simulations to improve the forming process and optimise the vial’s geometry — within ISO tolerances — at the handling and contact points in the heel area and at the lower ends of the side wall.
As a result, the vials can better withstand side compression and axial load during filling and transportation, reducing the risk of breakage and eliminating inherent stress in the glass.
Smooth and slick
Defects such as scratches and cracks on the surface of the glass are a frequent cause of breakage when mechanical load is applied during fill and finish or transportation, resulting in disruption to high-speed bulk production lines and increased manufacturing costs.
Furthermore, direct vial-to-vial contact on conventional bulk filling lines can create glass particles that may end up inside the container. EVERIC smooth vials have a coating on the outer surface that reduces the friction and allows the containers to run smoothly through the production line.
In developing this coating, SCHOTT placed great importance on making the vials abrasion-free and completely transparent. The layer does not impair the visual quality controls performed by the pharmaceutical company; furthermore, even critical pharmaceutical processes, such as washing and depyrogenation, can be done without alteration.
The coating not only protects against damage to the glass surface, but also significantly reduces particle formation. Tests have shown that the coefficient of friction of coated vials is approximately 56% lower than that of vials without a coating.
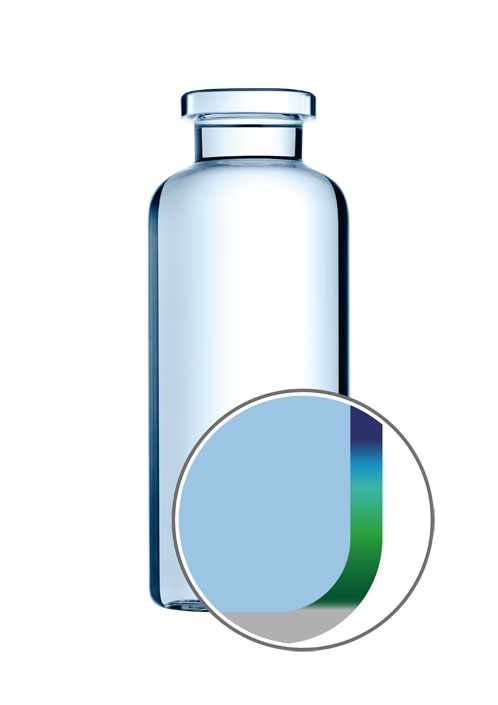
EVERIC strong
Customers can choose between two coatings — silicone- or silane-based — both of which show a hydrophobic surface behaviour. The silicone-based coating is applied via dipping and adheres to the glass surface by heat treatment.
Widely used as a gliding agent, silicone is typically relied on by high-speed lines for vaccines, generics and chemical drugs, for which avoiding interruptions and increasing yield are key.
EVERIC smooth: silane- or silicone-based
The silane-based coating is free from both nanoparticles and silicone, resulting in an overall homogenous surface energy. It is applied by covering the container with a molecular layer, which bonds covalently with the glass.
It is designed for companies filling high-value drugs such as biologics, which generally aim to exclude silicone from their facilities.
To demonstrate the advantages of EVERIC smooth vials in a practical setting, SCHOTT did a trial run of 17,000 vials with a silane-based outer coating on a commercial line and compared them with a similar number of uncoated SCHOTT TopLine quality containers.
The goal was to analyse the impact of the pharmaceutical filling line on container strength, prior to and after filling.
The 15 mL vials were filled with 12.25 mL of water for injection (WFI). Representative samples were selected after various process steps — washing, depyrogenation, crimping and final automated inspection — and subjected to a range of test procedures including burst pressure, diametral and axial compression load.
Additionally, the washing water and WFI were tested for residues from the coating material to assess any potential contamination risk. A 100% visual inspection of the processed vials took place to determine the effect of the coating on cosmetic quality.
The results demonstrated that EVERIC smooth greatly improves line performance by significantly reducing the number of final rejects and preserving the strength of the containers.
Although 235 uncoated vials were rejected because of visible defects, only five EVERIC smooth vials had to be discarded. This represents a 98% reduction in cosmetic defects.
In separate tests, 2R EVERIC strong and smooth vials were investigated for strength under real conditions against 2R vials of the highest quality. All vials were filled with WFI, sealed and crimped, then subjected to various tests according to ISO specifications: a burst test and a strength test involving axial and side pressure.
The goal was to test the extent to which the improved geometry and the outer coating made the vials more resistant to mechanical loads during the filling process.
With their optimised geometry, the EVERIC vials showed a significantly higher strength than the test sample vials in all three tests — both before and after the filling process.
Furthermore, the additional outer coating effectively reduced mechanically caused damage on the filling line (derived from glass-to-glass contact with other vials or glass-to-metal contact between the vials and the filling machine).
In the axial pressure test, the EVERIC strong and smooth vials demonstrated increased strength (by a factor of four) compared with the TopLine test vials. The side compression strength test showed an increased strength of 63% for the EVERIC vials at the end of the fill and finish process.
To manufacture pharmaceutical vials with the optimum strength to withstand the demands of today’s high-speed fill and finish lines, the obvious route might seem to be to produce a vial so hard that it prevents defects from occurring in the first place. But this would make little sense, because whenever a force acts, something has to give way.
That is why SCHOTT has taken a different route with its EVERIC concept. By introducing a modular concept for different features to improve the overall processing performance, SCHOTT offers pharmaceutical companies a choice of features that can be flexibly combined to meet individual requirements.

