From early clinical development and clinical trials to commercialisation, there are many obstacles that may stand in the way of an API realising its full economic potential. For example, as the manufacturing process is scaled-up, the original synthetic route may prove to be inefficient, unsustainable or too costly, whereas complicated or hazardous chemistries, or expensive key intermediates, may limit the ability to outsource or insource production, or may even make the development programme too uneconomical to continue.
Inlicensing late stage projects — from small biotech companies to large and medium sized pharma companies — often takes place at Phase IIb or even Phase III, but the limited resources and manufacturing experience of these small biotech companies mean that the processes are frequently not fully developed, very expensive, non-sustainable from an environmental standpoint or very difficult to scale-up to commercial quantities. However, a contract manufacturing organisation (CMO) such as Cambrex is able to identify opportunities to create significant technical advantages, thereby offering potential customers the benefits of substantial cost reductions, sustainable robust processes and additional intellectual property. When the company formed its dedicated Innovative Product Group in 2012, it had five clear objectives: to identify potential innovative opportunities; to develop the most cost-effective route; to identify and secure IP; to serve both branded and generic markets; and to supply commercially scalable processes.
However, for a CMO to become involved in such work requires significant resources as well as a high level of technical and commercial risk; and if the benefits are to outweigh these risks, initial project selection is crucial. The first key selection criterion is a technical evaluation. This will involve a comprehensive literature search and a freedom to operate (FTO) study to ensure that any new process development is non-infringing. A cost simulation will also need to be done, for both the existing manufacturing process and the proposed innovative process to assess the level of potential savings. The supply chain will need to be assessed and the new development and manufacturing strategy analysed.
A second key factor will be the assessment of the market: its value and the volumes of any potential API or key intermediate. It must also take into account any possibly competitive products that may be in the pipeline or could significantly influence market dynamics. The risks and rewards for each product are then evaluated.
Customer interaction is also important, particularly the sharing of clear cost targets and specifications. The customer needs to weigh up a cheaper route of manufacture against the cost and regulatory consequences of change. A range of potential supply strategies and business models will need to be considered, such as the supply of the API or key intermediates or a licensing model.
For its part, the CMO will need to show a good reason for the customer to change from the current synthesis or supply chain — and this is primarily achieved by offering a significant reduction in the cost of goods (CoG). The Cambrex Innovative Product Group sets a target in this regard of a saving of at least $5 million or a 25% CoG reduction. Both process and capacity should also be considered when looking to improve the CoG. All these evaluations need to be done prior to any laboratory work being undertaken by the CMO to ensure that development time is focused on where the most significant cost savings can be achieved.
Once a project has been given the green light, a proof of concept laboratory study is done. This would typically focus on the critical step in the proposed new process and could take 40 full time equivalent (FTE) days to complete. If this proves successful, and the market and customer situation is unchanged, a proof of scalability study is performed, during which the CMO will work to develop a robust and commercially appropriate manufacturing process. Depending on the complexity of the project, this could take 200 FTE days or more. Cambrex evaluates approximately 40 projects a year, of which some 20 go on to the proof of concept stage; around half of these, progress to a proof of scalability study.
Case study: dronedarone intermediate
The way in which a project can give rise to significant innovation was demonstrated by the development of a cost-effective, non-infringing process for a dronedarone intermediate, highlighting how a radical improvement in the manufacturing process can be achieved. A first generation five-step process, using readily available raw materials, was developed: the halogenated solvent in the Friedel-Crafts acylation in step 4 was successfully replaced with 2-nitro toluene, and the process was telescoped through steps 1–4 (Figure 1).
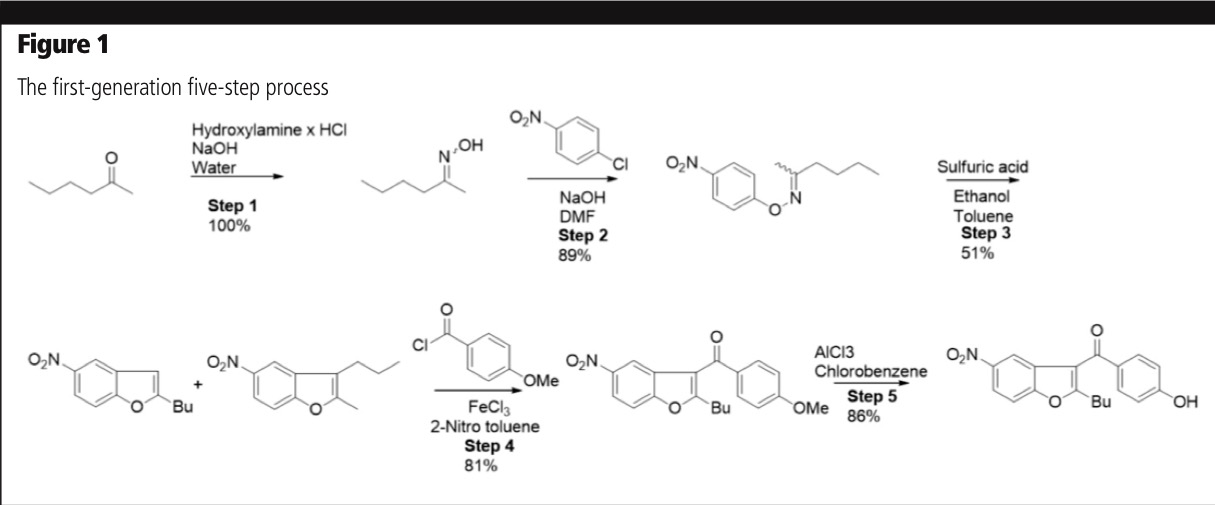
In principle, the process was scalable for industrial manufacture, but there were some limitations. The CoG was high and the overall yield was only 32%. The process was also non-sustainable, using 2.6 equivalents of aluminium chloride and chlorobenzene as a solvent to form methyl chloride as a by-product.
The five-step linear synthesis lacked flexibility and was not patentable owing to close prior art and the previous publication of similar chemistry. Furthermore, the process was not robust as out-of-spec batches could not be recrystallised to meet specification for a single impurity (0.10%).
Although the amount of alkylated by-product could be suppressed by running the process under reduced pressure or by passing a flow of nitrogen through the synthesis mixture, both procedures would increase halogenated waste to the atmosphere.
The project aimed to develop a second-generation process that would
- significantly reduce the COG
- use only standard equipment
- be a sustainable green process
- offer high capacity
- employ convergent synthesis
- be amenable to easy purification techniques
- be robust
- achieve high purity
- offer improved product properties with regard to electrostatic charges and dustiness.
Waste management was also developed in parallel with the process because of its significant influence on product throughput and overall cost. This was an important factor in facilitating a fast track scale-up from pilot to full-scale manufacturing. The second-generation process was a partly telescoped, four-step convergent synthesis, using easy-to-outsource intermediates that are well-defined solids. It achieved a yield of 65% from the A1–A2–C1 path, and a 56% yield from the B1–C1 path. Typical purity was >99.95% (Figure 2).
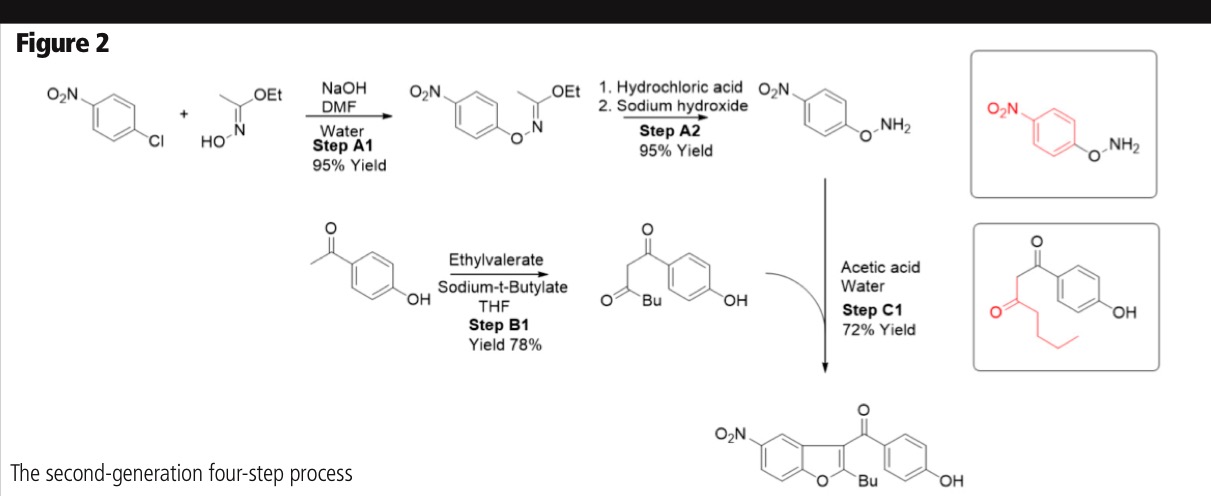
The shorter route, higher yield, lower waste disposal costs and less expensive raw materials meant that the CoG was reduced by 40%. The process was also more sustainable as there were no heavy metal catalysts or chlorinated solvents, the spent tetrahydrofuran solvent was returned to the supplier for work-up and the spent acetic acid was fermented to methane and used in energy production.
In addition, the process was robust, with more than 200 batches being manufactured in full scale, all within specification, using only standard glass-lined or stainless-steel reactors. The synthesis has also been patented (WO2009044143).
Understanding the first-generation process was key to the success of this project, according to Cambrex, whereas close collaboration with the customer and a clear appreciation of their needs throughout development was important in achieving goals. It is clear that for both originators and CMOs that there are significant and growing market opportunities to be gained from managing the lifecycles of APIs and key intermediates, but it is important to exercise caution and select projects with care. Innovation and process improvement can lead to a major reduction in the overall cost of goods; but, to achieve maximum benefits, both parties should be prepared to adopt a collaborative risk and cost-sharing approach.
