In this joint tabletting study by Laboratoire Pierre Fabre and Roquette, D Berthoumieu,* P Trannoy,* JF Cordoliani,* A François,** JM Hédin,** J Lis,** D Damour,** S Chesnoy** and P Lefèvre** highlight some of the ODT production benefits of a new excipient
Orally disintegrating tablets (ODTs) are dosage forms formulated in such a way as to improve a pharmaceutical product’s in vivo oral disintegration and dissolution rates. In order to achieve rapid disintegration rates, the tablet formula must provide a high porosity, low density and low hardness.1,2
The most common preparation methods used to produce tablets are: moulding, lyophilisation, freeze-drying and direct compression.3-7 The basic approach of developing ODTs via direct compression involves the blending of a filler, superdisintegrant, a lubricant and an active pharmaceutical ingredient (API) and then compressing the mixture.8–11
Mannitol is commonly used as a diluent or bulk excipient in the formulation of ODTs. A new generation of co-processed mannitol-based excipients has been developed by Roquette for the formulation of ODTs. For example, the latest – Pearlitol Flash – is a combination of mannitol and starch. Mannitol and starch are compliant with international pharmacopoeias and the combined ingredient is specifically designed to disintegrate rapidly, providing a smooth texture without the addition of superdisintegrant.
In the study presented here the physical properties of Pearlitol Flash were evaluated through a series of tests that revealed the impact of different compression parameters (pre compression and compression forces, tabletting speed) on tablet properties while using two different types of conventional tablet presses.
The materials used in this study were Pearlitol Flash (a combination of mannitol and starch from Roquette) and magnesium stearate as a lubricant (purchased from Bärlocher).
The powder properties, such as bulk and tapped density, angle of repose (using Hosokawa equipment), the flow rate through an orifice (using FloDex), the Hausner ratio and the Carr index, were all determined according to US Pharmacopeia (USP) and European Pharmacopeia (Ph. Eur) methods in order to characterise the physical properties of Pearlitol Flash.
Particle size distribution (sieving), loss on drying (IR method, Mettler LP16) and sorption of water (Dynamic Vapour of Sorption 20°C, DVS-1, Surface Measurement Systems LTD) were also evaluated. The Scanning Electron Microscopy (SEM) was performed using a Quanta 200FEG.
ODT preparation
Placebo ODTs were made by blending Pearlitol Flash and the lubricant in a Turbula T2C blender for five minutes. The tablets were prepared by direct compression using two different single punch tabletting machines: a Korsch XP1 press using 13mm flat bevelled-edge punches and set to achieve the same tablet weight (500mg) while increasing compression force (5–25kN); and a Stylcam simulator using 8mm convex punches (R8). In this case the press was set to achieve the same tablet weight (150mg) while increasing compression force (10–20 kN). The Stylcam was set to simulate a rotary press (Fette P1200) at a production rate of 72,000 tablets/hour (h).
The Stylcam Simulator is a single-punch press used to duplicate and analyse pre-compression, compression and ejection phases in industrial tablet production presses under identical conditions to those of a unit producing up to 300,000 tablets/h.
The amount of magnesium stearate used was adapted to the size of the tablet (see Table 1).
| Table 1: Formulas | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Formula (% w/w) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pearlitol Flash | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Magnesium stearate | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Table 2: Physical properties of Pearlitol Flash powder | |
| Bulk density (g/mL) | 0.527 |
| Tapped density (g/mL) | 0.631 |
| Flowability (g/sec) | 20 |
| Flodex (mm) | 4 |
| Angle of repose (°) | 40 |
| Haussner ratio | 1.19 |
| Carr index (%) | 16.5 |
| Loss on Drying (%) | 1.1 |
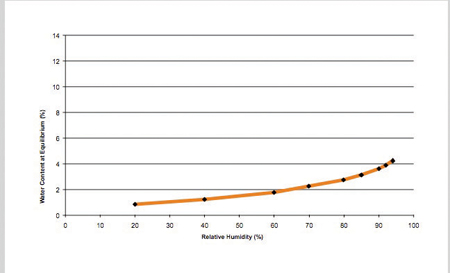
Figure 2: Dynamic vapour sorption isotherm of Pearlitol Flash at 20°C
The tests carried out using varying compression settings with the Korsch XP1 provided an insight into the influence of compression force on ODT tablet performance.
Pearlitol Flash was compacted at forces of 10, 15, 20 and 25kN. At 5kN the cohesion of the tablets was too low. Then the hardness of the tablets increased with the increased compression force. The 15kN compression force was sufficient to obtain tablets with acceptable hardness, friability and disintegration times (see Figures 3 and 4, and Table 3).
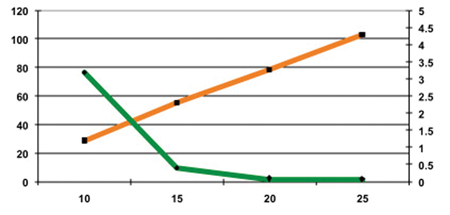
Figure 3: Hardness and friability as a function of compression force
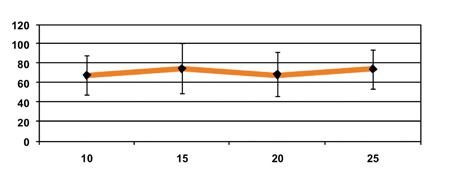
Figure 4: Disintegration time as a function of compression force
Surprisingly, the disintegration time was not significantly affected by the increase of the compression force and the tablet hardness. Generally, an increase in the compression force has a direct effect of lowering the disintegration time as it increases the hardness of the tablet and decreases its porosity. In ODTs, the absence of this interdependence between the disintegration time and the compression force has not previously been described.
tabletting speeds
To show the influence of tabletting speed on ODT performance, tablets were produced at 20, 40 and 60 tablets/min. No significant impact on the tablet properties was observed while increasing tabletting speed (20–60 tablets/min) (Table 4 and Figure 5). Tablet mass uniformity met the requirements of the Ph. Eur (m ± 5%). The hardness remained quite constant, while the disintegration time was lower but not statistically different for the higher tabletting speed. For all tabletting speeds, the increase in speed resulted in an increase of ejection force, but it still remained very low (under 230N). The low values for the ejection forces (Tables 3 and 4) confirm that 0.4% of magnesium stearate is enough to lubricate Pearlitol Flash.
| Table 3: Physical properties of Pearlitol flash tablets (mean ± SD) | ||||
| Compression Force (kN) | ||||
| Weight (mg) | ||||
| Ejection force (N) | ||||
| Hardness (N) | ||||
| Friability (%) | ||||
| Density) | ||||
| Porosity (%) | ||||
| Disintegration time | ||||
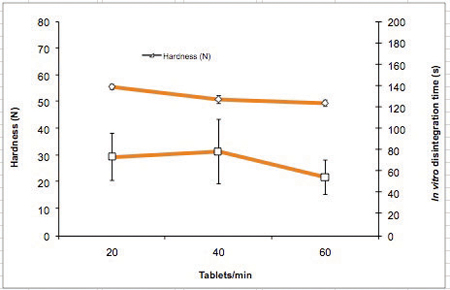
Figure 5: Hardness and in vitro disintegration time as a function of tabletting speed (compression force 15kN)
Table 4 shows the tabletting speed study which includes the compression and tablet data for a 15kN compression force (mean ± SD). Figure 5 also shows the hardness and in vitro disintegration time as a function of tabletting speed (compression force 15kN).
| Table 4. Tabletting speed study – Compression and tablet data - results shown for a 15kN compression force (mean ± SD) | |||
| Tabletting speed (tablets/min) | |||
| Weight (mg) | |||
| Ejection force (N) | |||
| Hardness (N) | |||
| Friability (%) | |||
| Density | |||
| Disintegration time | |||
The impact of compression settings on tablet properties were also studied using a Stylcam. This study was conducted to find the optimal tabletting parameters for the formulation of small (8mm) convex tablets of 150mg at an industrial level (Figure 6). The Stylcam was set to simulate a rotary press (Fette P1200) at a production rate of 72,000 tablets/h.
Among all the parameters that could be evaluated, the pre-compression and compression forces were the ones selected in addition to the impact of production rate.
pre-compression influence
The influence of a pre-compression force on ODT performance can be seen in Figure 6, where the hardness and friability are shown as a function of pre-compression force (compression force 10kN). Pre-compression forces of 0, 1.8, 3.2, 6.3 and 8.8kN were applied followed by a compression force of 10kN.
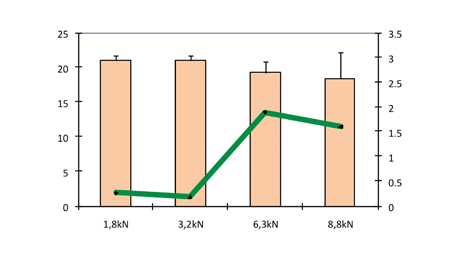
Figure 6: Hardness and friability as a function of pre-compression force (compression force 10kN)
If no pre-compression force was applied, there was systematic occurrence of capping. This phenomenon was greatly reduced by the pre-compression step, which contributed to removing the air inside the powder. A pre-compression force from 1.8 to 3.2kN was sufficient to prevent the occurrence of capping. Higher pre-compression forces above 6kN generated a new capping.
The increase in compression force shown in Figure 7 did not result in any increase in hardness. Capping has been observed to occur above a compression force of 16kN. Similar to the results obtained in the study conducted on larger sizes of tablet, in this study there was no effect by the compression force on disintegration time (Figure 8).
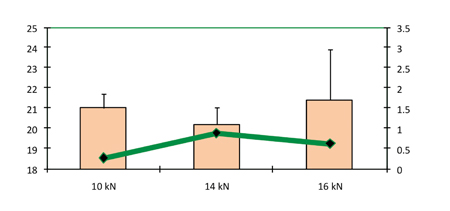
Figure 7: Hardness and friability as a function of compression force (pre-compression force 1.5kN)
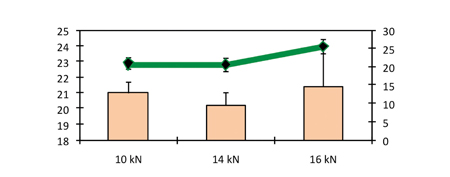
Figure 8: Hardness and in vitro disintegration time as a function of compression force (pre-compression force 1.5kN)
We can confirm here that the mechanisms underlying the disintegration process with Pearlitol Flash are different from those reported with ODT disintegration.1,2 It seems that the mechanism of disintegration is not predominantly driven by the porosity of the tablet (Table 3). We hypothesise that the wettability properties of the mannitol and starch compound play a major role resulting in a quick absorption of water by the tablet.
Figure 8 shows the hardness and in vitro disintegration time as a function of compression force for a pre-compression force of 1.5kN.
production rates
Figure 9 shows the hardness and friability as a function of production rate using a pre- compression force of 1.8kN and compression force of 10kN. Production rates of 28,800, 86,400, 120,000 and 144,000 tablets/h were evaluated.
At speeds of up to 120,000 tablets/h, there was practically no influence by production rate on the hardness and friability of the tablets. At 144,000 tablets/h capping was generated, reflected by the high variations in friability and disintegration times. The production rate had no influence on the disintegration time of tablets. This can be seen in Figure 10, which plots the hardness and in vitro disintegration time as a function of production rate using a pre-compression force of 1.8kN and compression force of 10kN.
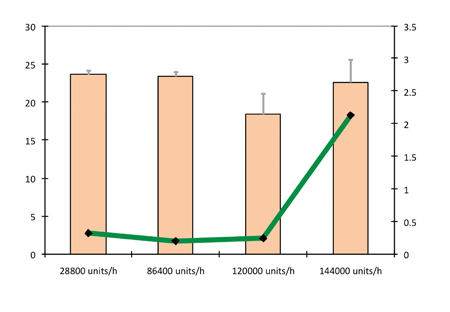
Figure 9: Hardness and friability as a function of production rate (pre-compression force 1.8kN and compression force 10kN)
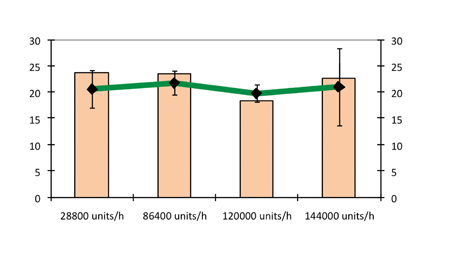
Figure 10: Hardness and in vitro disintegration time as a function of production rate (pre-compression force 1.8kN and compression force 10kN)
In conclusion, Pearlitol Flash has been developed as a self-disintegrating mannitol compound for the formulation of ODTs by direct compression. Irrespective of what size of tablet was chosen (13mm or 8mm), mechanically stable tablets were obtained for compression forces of around 14–15kN. The Stylcam simulator has allowed us to find the optimal settings for small sized tablets with a convex shape. A pre-compression step has been identified as necessary to limit the occurrence of capping. Thanks to the specific composition of Pearlitol Flash, only a very low level of lubricant is required (approximately 0.4–0.5%).
This study also revealed that the disintegration time could not be correlated to the compression force, nor the hardness or the tabletting speed. To conclude, Pearlitol Flash fulfils the requirement of excipients used to formulate ODTs in terms of flowability, hygroscopicity, compactibility, and disintegration.
footnotes
* Laboratoire Pierre Fabre, Ramonville, France
** Roquette Frères, 62080 Lestrem Cedex, France
references
1. Zhao N., Augsburger L.L. AAPS PharmSciTech 6(1): Article 19 (2005)
2. Carter J.C. Pharmaceutical Canada, 3, 2, 1-4 (2002)
3. Harmon T. Tablets & Capsules, 1-5 (2006)
4. Bhowmik D., Krishnakanth C.B., Chandira P.R.M. J. Chem. Pharm. Res., 1(1):163-177 (2009)
5. Shaikh S., Khirsagar R.V., Quazi. Int. J. Pharmacy Pharm. Sci., 2(3) 9-15 (2010)
6. Gupta A, Mishra AK, Gupta V., Bansal P., Singh R. Singh AK. Int. J. Pharm. Bio. Arch., 1(1):1-10 (2010)
7. Wagh M.A., Dilip K.P., salunkhe K.S., Chavan N.V., Daga V.R. Int. J. Drug Del, 2:98-107 (2010)
8. Balasubramaniam J and Bee T. Pharm. Tech. (Suppl. April 2009)
9. Singh S.S., Mishra D.N., Jassal R., Soni P. Asia. J. Pharm. Clin. Res., 2(3):74-82 (2009)
10. Shahi S.R., Agrawal G.R., Shinde N.V., Shaikh S.A., Shaikh S.S., Somani V.G., Shamkuvar P.B., Kale M.A. Rasayan J. Chem, 1(2):292-300 (2008)
11. Chaudhary S.A., Chaudhary A.B., Mehta T.A. Int. J. Res. Pharm. Sci., 1(2):103-107 (2010)
12. Yoshinari T., Forbes R.T., York P. Kawashima Y. Int. J. Pharm., 247: 69-77 (2002)
You may also like
Roquette's Low Nitrite METHOCEL HPMC wins NIE New Ingredient award
Read moreTrending Articles
-
You need to be a subscriber to read this article.
Click here to find out more.
