The implementation of continuous manufacturing (CM) for the production of oral solid dosage (OSD) products has demonstrated a number of key benefits to the pharmaceutical industry, including reduced development times, increased flexibility and a greater level of agility to deal with process changes.
Yet, as the continuous processing of pharmaceuticals has moved out of the laboratory and into production environments, questions regarding the capability of the equipment to reliably and compliantly produce high quality product for long periods of time have been raised.
To examine these issues, GEA, in collaboration with MSD, a tradename of Merck & Co., Inc. (Kenilworth, NJ, US), recently conducted a successful robustness run on GEA’s ConsiGma CDC 50 Continuous Direct Compression system for a period of 120 hours. The positive outcome highlights the suitability of CM technology for the efficient completion of campaigns that formerly involved longer and more complex batch processes.
CDC 50 overview
The ConsiGma CDC is a compact, all-in-one, tablet production line for direct compression (DC) formulations, meaning that the dispensing area, blending area and intermediate storage requirements are a thing of the past. Up to six ingredients can be fed separately via specially developed loss-in-weight (LIW) feeders and, thanks to the integration of the feeding, blending and compression operations, segregation issues have been eliminated.
Proven blending technology results in minimised start-up and shutdown losses and the unique GEA Air Compensator compression technology offers extended and controlled dwell times, accurate weight management and minimal variability in critical tablet quality attributes (an automatic tablet sampling and analysis system is also available).
This article outlines the operational advantages of the GEA ConsiGma CDC 50 during a prolonged trial, with a detailed assessment of the representative 120-hour commercial run. Aspects covered will include the following:
- process effectiveness: yield of run, losses from the system and reasons for rejection during the campaign
- implementation of controls: how a multilayered control philosophy can ensure product quality
- overall equipment effectiveness: a review of the cleaning philosophy of the CDC 50 and how this would translate to commercial production and system turnaround time.
The trial run was monitored using a suite of tools, including the high-frequency measurement of more than 100 process and environmental parameters, such as spectroscopic analysis of the powder blend and physical testing of the finished product; soft sensors were used to predict critical quality attributes (CQAs).
Particularly noteworthy was the use of an advanced process model to predict tablet potency and accept or reject tablets at the end of the production line. This advanced control strategy was developed by characterising the residence time distribution (RTD) of each blending stage using near infrared (NIR) spectroscopy techniques with a GEA Lighthouse Probe embedded within the production line.
Project aims
The aim of the experiment was to run the CDC 50 for a target time of 120 hours, recording any encountered issues during prolonged operation at a nominal flow rate of 50 kg/h. The objective was to manufacture 6000 kg of finished product.
A formulation that challenged the system but is representative of common DC formulations was used and factors such as build-up, material loss and the long-term performance of the different subunits was assessed.
It was decided to use a commercial control strategy using online PAT, process modelling and material tracking to ensure the quality of the final product; samples of the final tablets were taken periodically to measure standard parameters using at-line in-process control methods.
Results
An overall summary of the run is shown in Figure 1. By the end of the 120-hour trial, more than 15 million tablets had been made using approximately 6200 kg of raw material in a single production area. Importantly, final analysis indicated that the campaign length could be increased even further and run for longer.
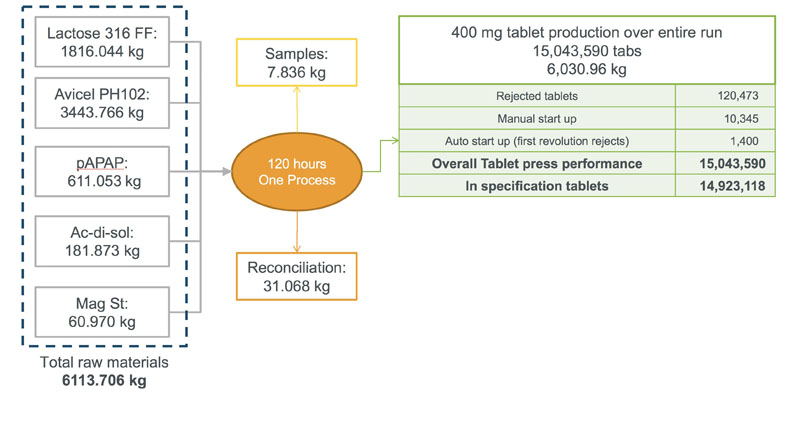
Figure 1: An overall summary of the run
Benchmarking against a typical batch process, producing the same quantity of tablets would have required 10 separate campaigns and taken a team of operators working in parallel in three production areas approximately 5–7 days.
Reconciliation: At the end of the run, all possible losses of dust from the system were captured and recorded. In total, 31 kg of material was recovered, with more than 55% of that being unused excipients in the manual handling system.
Press controls: Every 15 minutes, a sample of 10 tablets was taken and tested on an automated tablet tester. Plotting the results as a process capability (within the ±4% control limits set), the overall process capability of the entire run was calculated to be 2.06 — a Six Sigma controlled process (Figure 2).
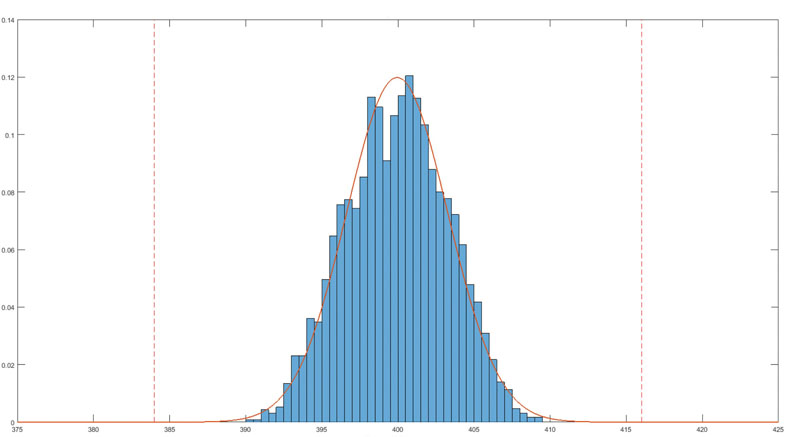
Figure 2: The overall process capability was calculated to be 2.06 (Six Sigma)
Control response during run: The rate of diversion (per 12-hour slot) was monitored throughout the experiment. The majority of diversions occurred at the start of the run when there was a mechanical issue that required intervention (first 36 hours).
The last 86 hours of the run had an average reject rate of just 5300 tablets per 12 hours. Overall, the majority of diversions were because of the residence time distribution (RTD) system tracking disturbances and deeming them to be out of specification.
RTD model: The control model on the CDC 50 used during the run converts the individual component mass flow data into predictions of the blend potency at key stages of the system. The system uses configurable RTDs of the different unit operations to track concentration variability. The RTD model was calibrated with a system disturbance at the start of the 120-hour run (clean) and at the end (dirty).
The model “spike” responses — measured using the NIR system and key data — show virtually no change in the system RTD during the entire run, which indicates a high level of confidence that the RTD prediction (Figure 3) for the whole campaign is accurate.
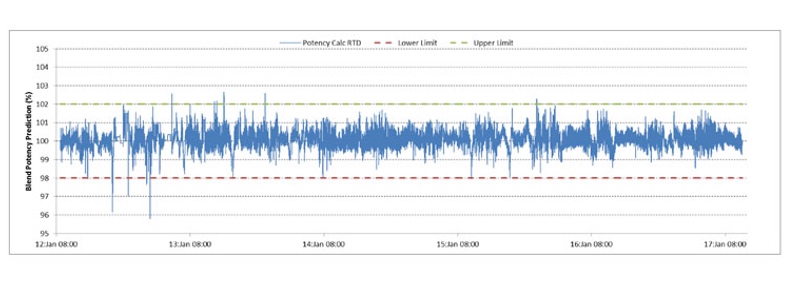
Figure 3: The model spike responses show virtually no change in the system RTD during the entire 120-hour run
HOTSWAP cleaning philosophy: As areas of the critical turnaround path for the CDC 50 can be set up as HOTSWAP parts, it’s comparatively easy to have standby parts clean and ready to go. This offers benefits with systems that require extra time because of mechanical complexity and/or sensitive technologies (feeder assemblies, for example) that require careful handling, alignment and calibration to ensure correct operation.
Using HOTSWAP principles can increase the productivity of a CDC 50. Using a shift format of 12 h x 5 days and conventional changeover functionality, it is theoretically possible to make 300 million tablets per year (100 million each of 100 mg, 200 mg and 400 mg), representing an OEE of 55.0%. By contrast, 403 million tablets per year can be produced using HOTSWAP principles (134.4 million of each dose size), representing a 31.4% increase in productivity and an OEE of 72.5% (data not shown).
In conclusion
This study shows the capability of the CDC 50 to manufacture a representative direct compression tablet under production conditions. The process was able to manufacture 15 million tablets, with a final yield of 99.2%.
Using a commercial control strategy, the process was monitored and controlled in real-time, with multiple layers of control working together to ensure the final product quality (NIR for blend and content uniformity, RTD modelling and compression-specific control of weight, thickness and hardness).
Weight control, for example, from the GEA tablet press is shown to have a high process capability (Six Sigma) during the entire 120-hour run. Plus, using HOTSWAP principles, the turnaround time of the CDC 50 can be dramatically reduced, increasing the available production time and improving the overall OEE of such a system.
Summary statistics revealed that greater than 99.5% of the production met or exceeded CQA specifications, and fewer than 0.5% of the tablets were out-of-specification, providing a solid body of proof that the technology is eminently suitable for the efficient completion of campaigns that formerly involved longer and more complex batch processes.
Additionally, a production uptime of 97% demonstrated reliability that exceeded industry benchmarks, and reconciliation above 99% showed good accountability of raw material and finished product. In conclusion, the 120-hour campaign achieved the goal of demonstrating the capability of continuous manufacturing to reliably, flexibly and compliantly produce high quality drug products on demand.

