Recombinant proteins are produced using genetic engineering techniques. This article explores their purification, the importance of effective concentration, and how it can be achieved safely.
Why produce proteins recombinantly?
Most proteins are not naturally produced in a form or in quantities that allow easy purification. Recombinant protein production overcomes the limitations of naturally produced proteins by making cells synthesise specific proteins in amounts that can be purified for use in fundamental research or for industrial and therapeutic applications.
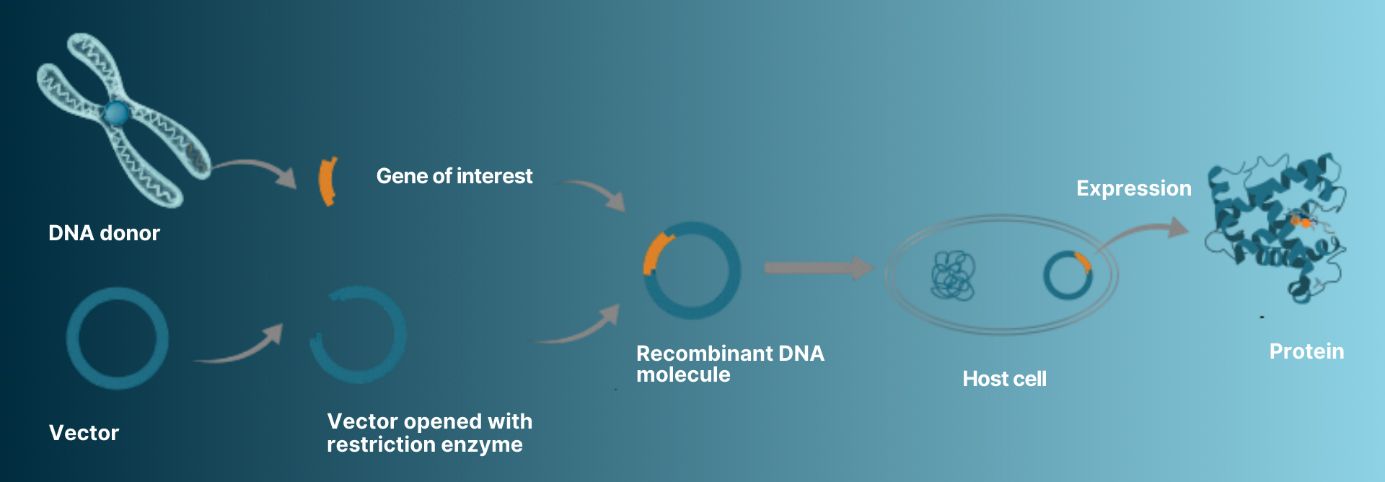
Recombinant protein expression
Recombinant protein purification:
Recombinant protein purification is a fundamental biotechnology process that isolates a target protein from a complex biological mixture to obtain it in a highly pure and functional form. Effective purification removes host cell debris, nucleic acids, endotoxins and other impurities while preserving the protein's native conformation and biological activity; however, poor purification may lead to protein aggregation or loss of function.
The purity needed depends on its end use. For proteins used in research, 90–95% purity may be sufficient, but for proteins used for pharmaceutical applications, much higher purity levels of up to 99.99% must be achieved.
The protein purification process typically consists of several steps, each of which involves increasing protein purity while maintaining structural integrity. The stages are:
- Sample collection and preparation
- Cell lysis and protein extraction
- Solubilisation and stabilisation
- Purification
- Concentration/storage
Depending on the protein and the intended application, extra processes may be necessary to refine and validate protein quality.
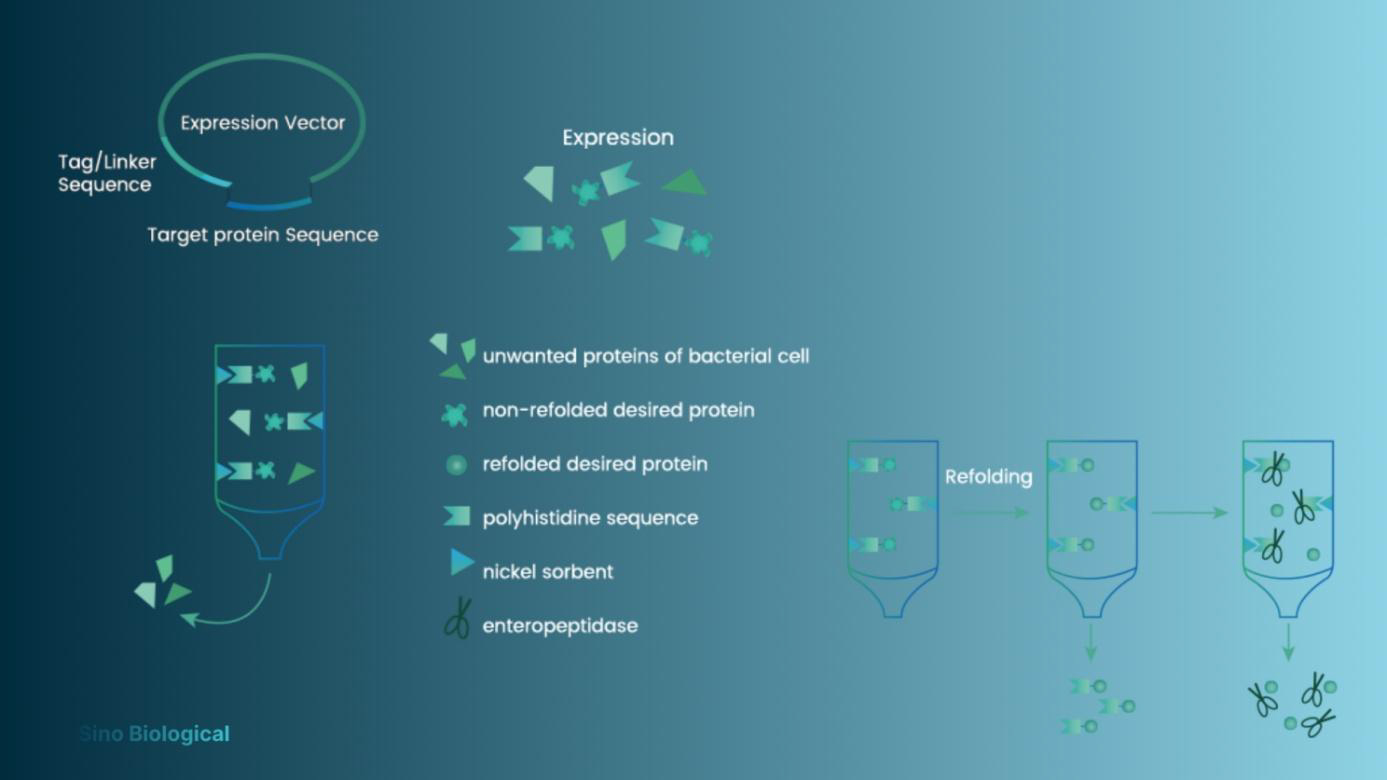
Affinity Tag Purification
Recombinant protein purification in drug discovery:
Recombinant proteins can be used in drug discovery both as targets for high-throughput screening and as therapeutic drug candidates. They also contribute to structural biology studies to guide rational drug design. As such, protein purification helps in the study of protein structure, function and interactions. Post-purification, proteins are frequently concentrated in high quantities.
Why concentrate recombinant proteins?
This process is vital for preparing the protein for subsequent applications, and concentration plays a key role in keeping proteins stable in solution. Proteins need to be stored at a high level of concentration, at least 1 mg/ml, as dilute protein solutions (< 1 mg/ml) are more prone to inactivation and loss as a result of low-level binding to the storage tube.
Several methods are available for protein concentration. One is:
Centrifugal vacuum concentration:
For laboratory-scale protein concentration, this method removes the solvent from the supernatant under vacuum so that the boiling temperature remains below the protein’s denaturation threshold. This approach can significantly improve both the yield and the activity of the protein after concentration.
In practice, centrifugal vacuum concentration can be achieved using instruments such as the centrifugal evaporator EZ-2 by Genevac, which can handle a wide range of samples, including proteins, enabling precise solvent removal without compromising sample integrity and making it an essential instrument in many laboratories.

EZ-2 Elite
There are two main challenges when concentrating proteins using centrifugal evaporators: overheating and cross-contamination. The EZ-2 centrifugal evaporator is designed to address these issues, as detailed below:
Overheating:
Heat energy is necessary to replace that lost as latent heat of evaporation in the boiling sample. As the solvent boils, it loses heat energy and cools itself and the container. This slows evaporation, and thus energy must be directed into the drying sample to replace that which is lost if a continuous evaporation rate is to be maintained. Infrared heater lamps, which are a development of halogen lamp technology, are very good at providing the necessary heat flow. However, they can sometimes be too efficient, leading to overheating of a sample that has already reached dryness. This is extremely undesirable where proteins and peptides are concerned, since they are thermally labile and easily damaged by temperatures above 40°C. To prevent this situation, it is necessary to measure the temperature of the sample as it spins.
This issue is overcome in the EZ-2 by using a finely tuned IR pyrometer combined with sturdy, solid aluminium sample holders. The non-contact sensor measures the surface temperature of the aluminium as it passes by and is accurate to less than 2.5°C, which is acceptable for this application. Because heat flow through the aluminium sample block is uniform, and because the instrument can control the heat flow to the samples by switching the IR lamps on or off, it is possible to deduce the actual sample temperature from these data points using a simple algorithm. In this way, using SampleShield™ temperature control technology, the EZ-2 allows scientists to preselect a sample protection temperature suitable for biology applications, normally 35 or 40°C.
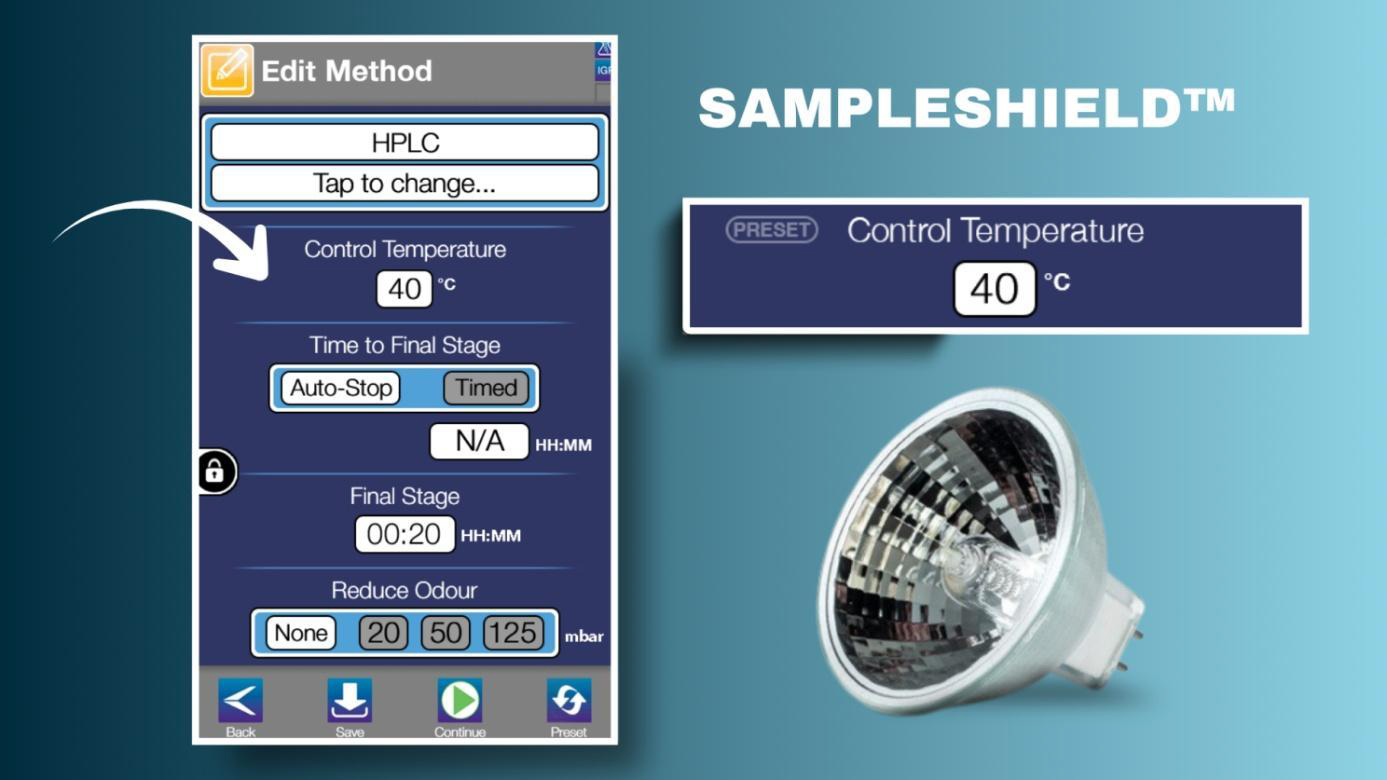
SampleShield temperature control technology
Cross-contamination:
When using microplates with highly sensitive samples such as proteins or peptides, there is a risk of cross-contamination during the concentration stage. Concentrating large biomolecules in these microplates is not straightforward, and this is where the EZ-2 centrifugal evaporation technology provides a solution.
In a conventional evaporator, the plates spin at a speed that produces 250 to 300 xg, and this level of g-force is insufficient to entirely prevent cross-contamination within the plate. Contamination arises if samples begin to bump during the evaporation process.
It can be eliminated by the use of the DriPure™ anti-bumping system and EZ-2. With DriPure™ enabled, the vacuum is gently ramped down over a period of approximately 30 minutes. At the same time, the applied g-force is increased to over 450 g to prevent bumping by accentuating the boiling point/depth gradient and concentrating all the “ready to boil” solvent near the surface. This also creates active convection and ensures good mixing so that temperature gradients do not arise, which could cause chaotic mixing of areas of liquid at dissimilar temperatures. DriPure also ensures that if material is eventually ejected from the liquid surface, it is kept within the plate well.

EZ-2 Elite
The EZ-2 centrifugal evaporator can be used to concentrate recombinant proteins. The important step is to select the correct method. Alternatively, we can help determine which method best suits your sample and process requirements.
References:
Protein Expression and Purification Series- bio-rad
Recombinant Protein Purification: Definition, Methods, Steps, and More - Sino Biological
Protein purification: Methods, techniques, and applications -abcam
How to Choose the Right Recombinant Protein for Your Experimental Model
Protecting activity of desiccated enzymes
Choosing the Right Centrifugal Evaporator for Your Lab's Needs
The Importance of Controlled Concentration and Drying in MALDI-TOF Applications
Genevac Evaporators and Concentrator Applications
Genevac EZ-2 4.0 Series Centrifugal Evaporators | SP Industries Inc.

